Tumor Mutational Burden & MSI
OnkoGenetiks™ Tumor Mutational Burden analysis determines the number of somatic mutations in the tumor genome and serves as a potential biomarker for cancer therapy.

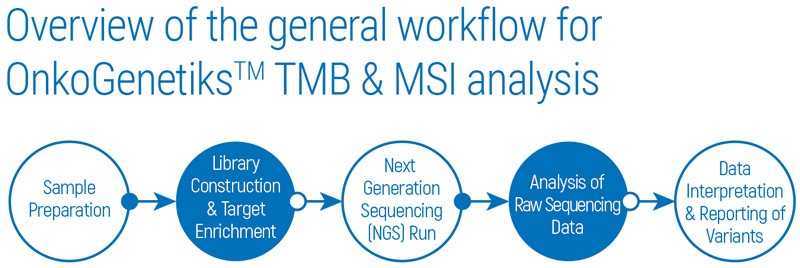
The overall procedure starts with DNA extraction from FFPE tissue and blood samples. After library construction and target enrichment with QIAseq Targeted DNA Panels, NGS is performed by using Illumina NextSeq 550Dx sequencing platform. Analysis of raw sequencing data is carried out and detected variants can be interpreted with the Ingenuity®Variant Analysis (IVA) or QIAGEN Clinical Insight (QCI™) tools.
Microsatellite Instability (MSI) panel contains 7 markers corresponding to the MMR (Mismatch Repair) genes. MSI analysis is performed with ABI 3500xL Genetic Analyzer device and results are computed using the GeneMapper software.
Checkpoint inhibitor (CPI) therapy, which is a form of cancer immunotherapy, has demonstrated a remarkable clinical benefit in recent years for many cancer types such as skin, lung, bladder, kidney and in tumors having deficiency of MMR (Mismatch Repair).
Cancer immunotherapy emerged to be a very promising and powerful tool for the treatment of cancer. However, the ability to successfully identify patients who will benefit from this kind of therapy is still limited.
Tumor mutational burden (TMB), a measure of the number of somatic mutations per coding area of tumor genome, is a putative biomarker of response showing great promise in CPI and immunotherapy combination trials.

Non-synonymous mutations are changes in DNA that result in amino acid changes in the protein. These mutations have the potential to generate neoantigens recognized by the host immune system, leading to an antitumor immune response. Tumors with high mutation burden have the potential to generate a larger number of neoantigens, making them more immunogenic.
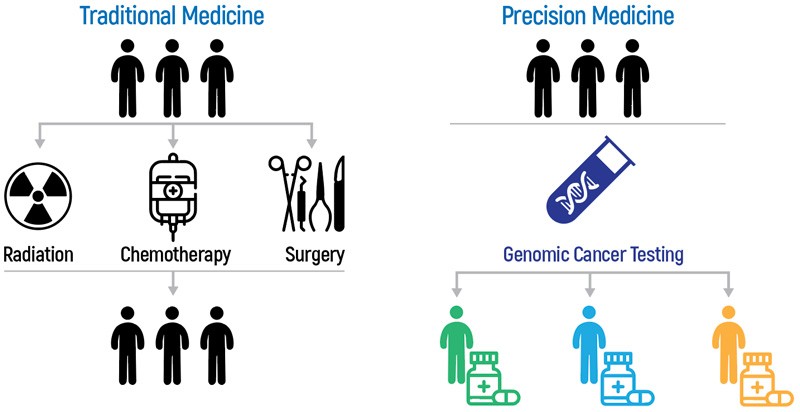
Precision medicine allows doctors to offer more individualized treatments to cancer patients based on their genetic features in order to obtain better efficacy.
Why perform TMB analysis?
• Only 20-40 % of cancer patients respond to treatments based on immunotherapy.
• Obtaining the knowledge of mutations in your cancer cells can help you and your physician to better understand and decide on your treatment options.
• High Tumor Mutational Burden (TMB) was found to be associated with better prognosis in patients with various cancer types undergoing immunotherapy treatments.
• Determining the Tumor Mutational Burden (TMB) and applying the appropriate treatment options can save important amount of time and cost.
Why OnkoGenetiksTM TMB?
• Includes 486 genes relevant to cancer and immune system.
• ~1.3 Mb of target DNA region is covered in total.
• High TMB confidence - at least 1 Mb target region has 100X or higher coverage.
• More than 16 years of QIAGEN experience and over 10 million biomedical findings.
• Access to over 1 million of unpublished variant-phenotype relationship with Qiagen bioinformatics solutions.
• Test performed in Turkey
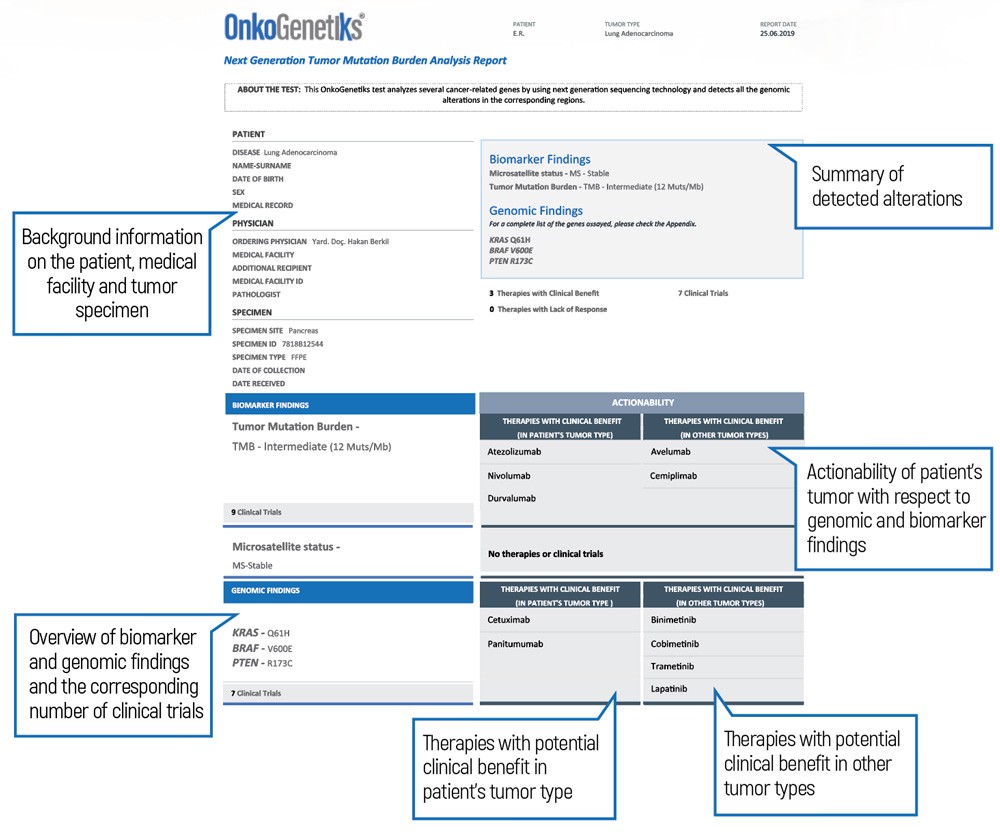
OnkoGenetiksTM Tumor Mutational Burden analysis reports
• Presented in an easily interpretable format in order to provide the highest possible clinical benefit.
• Your physician can have access to your tumor profiling information in a clear and comprehensible manner.
• A turnaround time of 14-21 days helps you to find the answers you seek as fast as possible.
OnkoGenetiksTM Likid
Next Generation Oncology Screening
Liquid biopsy for detection of somatic mutations in circulating cell-free tumor DNA (ctDNA) from a blood sample.
WHY LIQUID BIOPSY?
“Liquid biopsy” is a safe, non-invasive, highly sensitive and cost effective method of analyzing circulating tumor DNA (ctDNA) from the plasma of patients diagnosed with cancer or from individuals who may have cancer.
By analyzing ctDNA isolated from a patient’s blood, we can identify clinically relevant genomic alterations in ctDNA and match these alterations to targeted therapies and clinical trials.

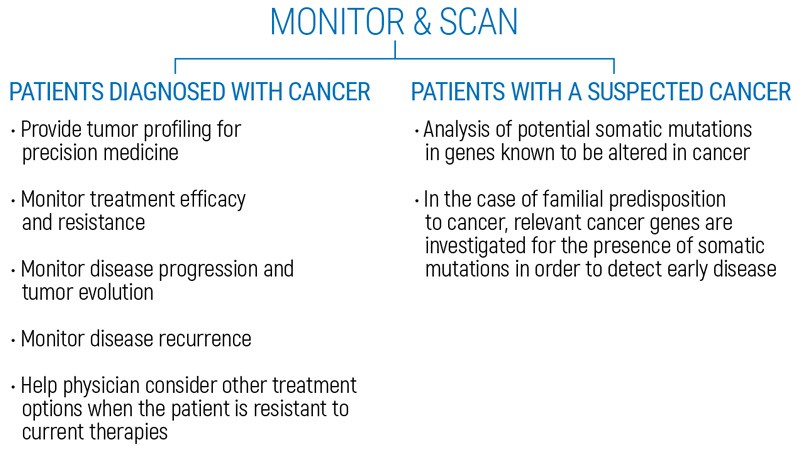
Identification of somatic mutations on circulating tumor DNA (ctDNA) for cancer monitoring or for early detection of cancer. OnkoGENETIKSTM Liquid Biopsy test is intended for patients who have been diagnosed with cancer or individuals with high risk for a suspected cancer.

Overview of the general workflow for OnkoGenetiksTM Liquid Biopsy Analysis
The overall procedure starts with DNA isolation from blood samples obtained in PAXgene Blood ccfDNA Tubes. After library construction (using OnkoGenetiksTM designed gene panel) and target enrichment steps, NGS is performed by using Illumina NextSeq 550Dx sequencing platform. Analysis of raw sequencing data is carried out and detected variants can be interpreted with the Ingenuity®Variant Analysis (IVA) or QIAGEN Clinical Insight (QCI™) tools.
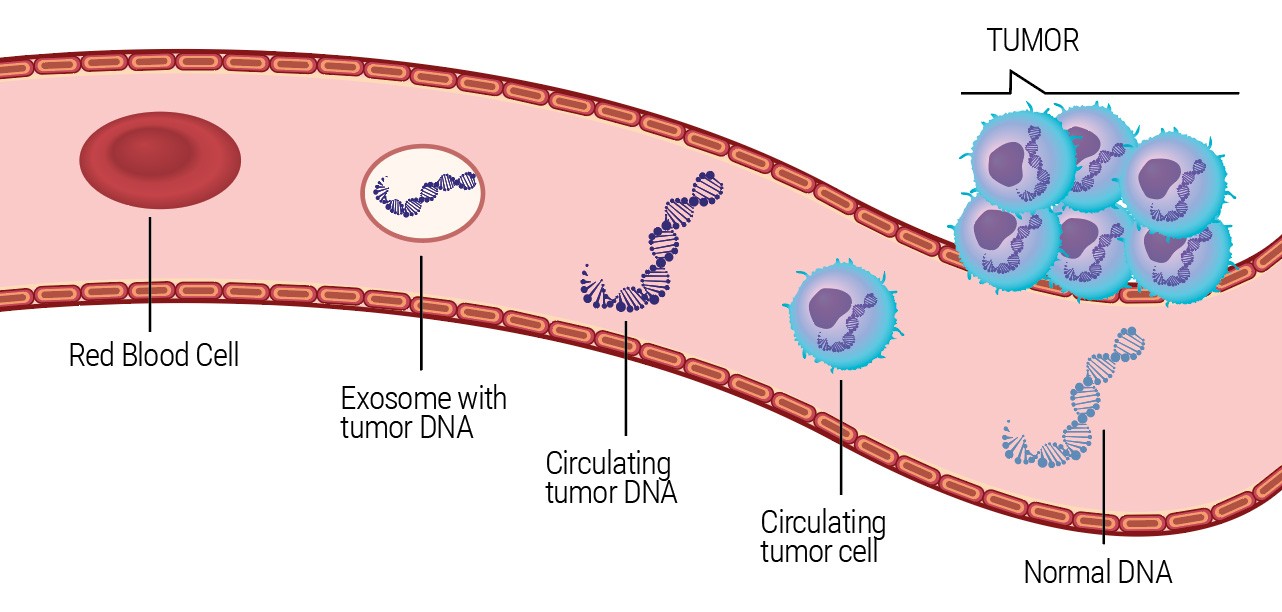
As healthy and diseased tissue interact with the bloodstream, they release both cellular and genetic material into the circulation. These materials are referred to as “cell-free” DNA (cfDNA), and circulating tumor cells (CTCs). Tumor DNA from these circulating tumor cells is termed as circulating tumor DNA (ctDNA).
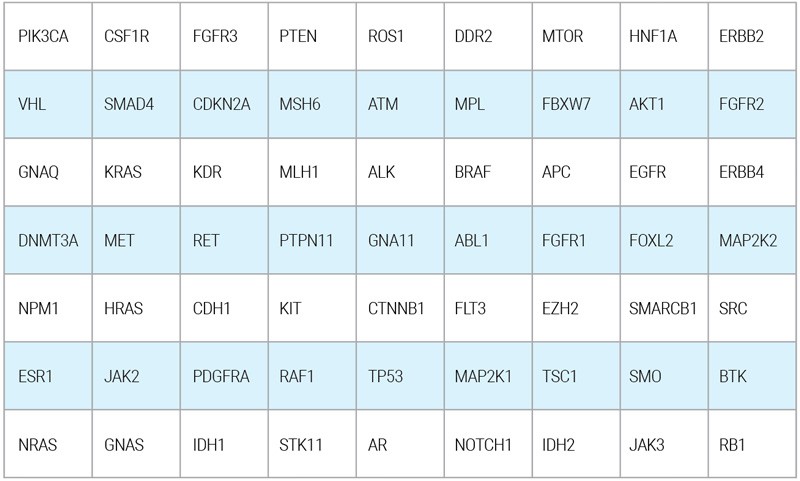
OnkoGENETIKSTM Liquid Biopsy gene panel covers a broad spectrum of high priority genes which are known to be most involved in cancer progression. The genes are stated as the following: (Hotspot regions are being analyzed for certain genes)

 Türkçe
Türkçe English
English عربي
عربي